Microgravity enables differentiated drug products
Formulation challenges limit access to key therapies for patients—processing in microgravity can be the solution.
Turning gravity “off” impacts material processing

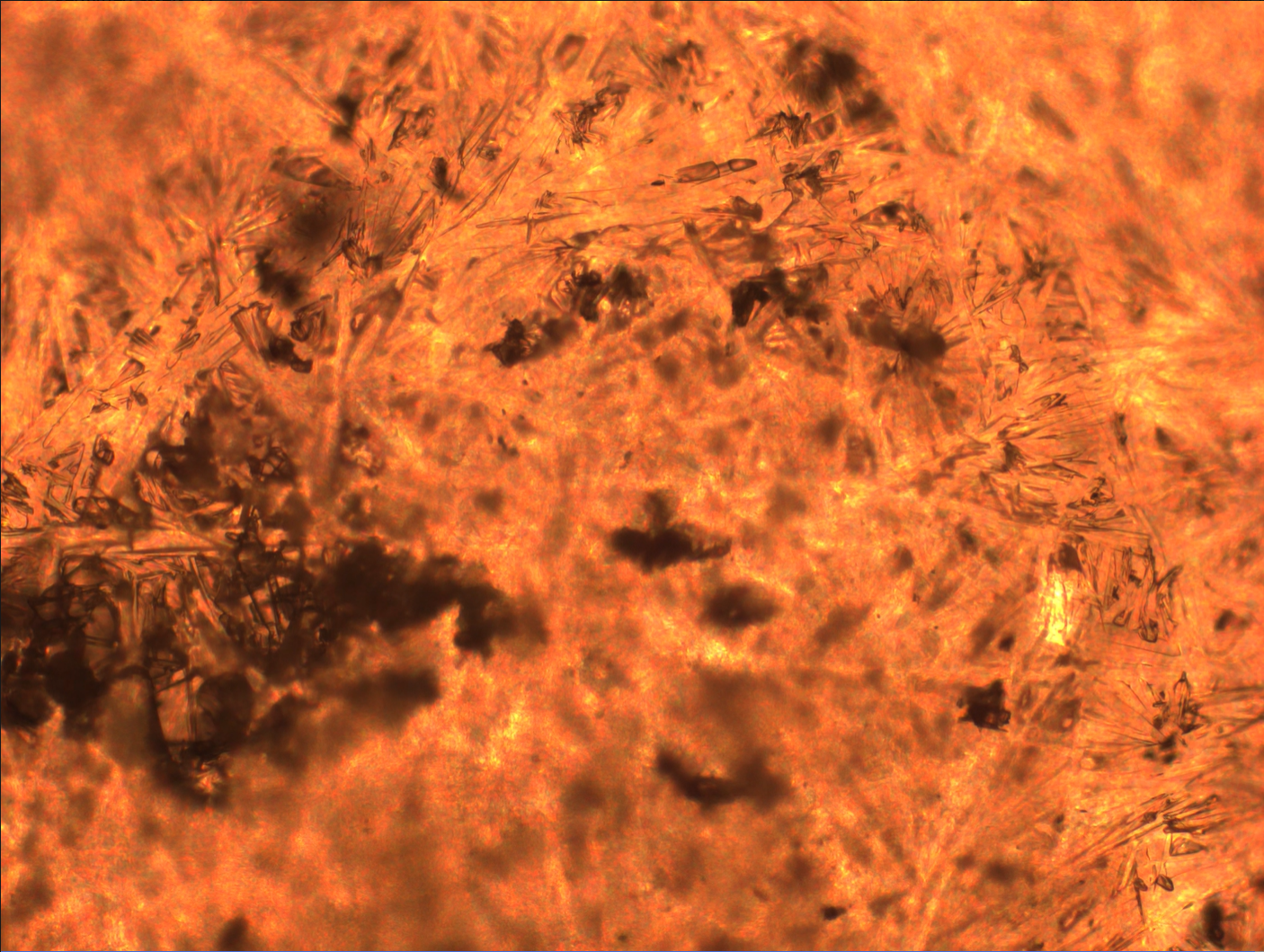
Microgravity suppresses convective currents and sedimentation, leading to more uniform crystals in space (right) than on Earth (left).
Processing materials in the near-weightless conditions found in space offers a unique environment that is impossible to replicate on Earth.
Gravity impacts many chemical processes, including how we make medicines. Microgravity suppresses convective currents, buoyancy, and sedimentation, and the resulting crystals are more uniform in size and structure.
When processing materials in orbit, the effects are “locked” into the material, before being brought back to Earth to be manufactured at scale to benefit patients.
Formulation
Varda’s microgravity platform provides a unique path to formulating small molecules and biologics to improve shelf-life, bioavailability, and the patient experience.
Crystallization
Reduced convection and sedimentation allows for particle size tuning and can result in the formation of large and ordered crystals for high-resolution X-ray structural determination or small and uniform particles to improve drug delivery.
Intellectual property
The unique environment of microgravity allows for the generation of enabling product and process IP for new chemical entities (NCEs) as well as life cycle management of marketed drugs.
Screening
Varda's extensive terrestrial crystallization lab includes a pharmaceutical hypergravity facility designed to accelerate development programs, building gravity-dependencies directly into drug substance development right here on Earth.
Gravity-enabled pharmaceutical development funnel
Assess feasibility and optimize terrestrial process
Varda conducts process characterization on custom and traditional crystallization hardware for small-volume screening (Crystal16, Technobis) as well as larger-scale automated reactors (EasyMax, Mettler Toledo) with complimentary process analytical technologies. Varda offers:
- Traditional solid state (XRPD, DSC, and TGA) and particle characterization (laser diffraction, dynamic image analysis).
- Crystallization process development, coupled with in-situ sensors (video microscopy, Raman, infrared) to probe and assess crystallization kinetics in real time to map process windows and ensure robust experimental design.

Use hypergravity to rapidly assess gravity’s impact to your therapeutic program
Terrestrial hypergravity testing unlocks commercial opportunities for in-space production. Crystallization experiments are conducted with custom or commercial off-the-shelf hardware compatible with Varda’s hypergravity platform to assess drug properties and performance across a range of gravity levels. Varda’s hypergravity platform allows us to:
- Perform drug development unit operations under hypergravity.
- Observe and quantify gravity-dependent drug product performance metrics as a function of gravity (size vs. gravity, polymorph ratio vs. gravity, and more).
- Develop process models which incorporate gravity as a variable to enable predicting or anticipating the impact of microgravity on drug properties or performance.
Hypergravity screening is a cost-effective way to derisk selection of pharmaceutical compounds and materials to identify those that are best suited for in-space development and greatly improve the success rate of microgravity studies.

Autonomous in-orbit process development with material return to Earth
Varda’s capsule allows process development in orbit with return of materials to Earth. To conduct testing in orbit, Varda handles each step for our customers:
- Small molecule and biologic process development and manufacturing capabilities
- Sample handling, preparation and loading for in-flight studies
- Full mission operations and experimental controls from Los Angeles, CA ground based facilities
- On Earth, sample recovery, handling and analysis

Comprehensive analysis of microgravity-produced materials
Once our customers’ sample returns to Earth, they can fully characterize all materials produced and recovered from microgravity studies with Varda or through external strategic partners.
Varda and its partners are equipped with a full suite of analytical testing capabilities:
- Diffraction: SCXRD, XRPD, T-XRPD and Synchrotron
- Thermochemistry: DCS, mDSC and TGA
- Spectroscopy: FTIR, NMR, Raman, UV-VIS
- Microscopy: SEM and TEM
- Others: Dissolution, DVS, HPLC, KF, LOD and PSD

Want to learn more about how microgravity improves drug formulation?
Thank you

Do you have a molecule that is impacted by gravity?
Varda’s hypergravity crystallization platform demonstrated the effect of gravity on nucleation and growth kinetics for a seeded cooling crystallization of L-histidine. The study illustrates the ability to use gravity as a variable to influence particle size distributions of small molecule therapeutics.
Please contact pharma@varda.com for microgravity crystallization information.